A world-first breakthrough could see COVID-19 blood test results returned in 20 minutes, with hospitals able to process 700 tests per hour.
Developed by researchers at Monash University’s Department of Chemical Engineering, the tool detects the presence of antibodies raised in response to COVID-19 infection.
The simple assay separates free cells from red blood cells, which have been found to cluster when containing COVID-19 antibodies. It is based on commonly used blood typing infrastructure that is already manufactured at scale.
Project leader Dr Simon Corrie runs the Nanosensor Engineering Lab at Monash University, which focuses on developing new biosensors and assays.
“There were lots of PCR [polymerase chain reaction]-based tests available, which is the swab test where people are detecting directly the genetic material in the virus which is indicative of current infection,” he told create.
“But we could see an opening for rapid lab based blood testing, because there you detect the antibodies that are raised by the immune system in response to the virus.”
Corrie and his team of researchers and PhD students were able to quickly mount the case for a targeted group of people to work exclusively on the project once Victoria state went into Stage 3 lockdown in March.
“We identified blood type tests as an already scalable, simple and widespread technology available all over the world, and we modified them ever so slightly to actually detect antibodies raised against the virus in patient blood,” he said.
How does it work?
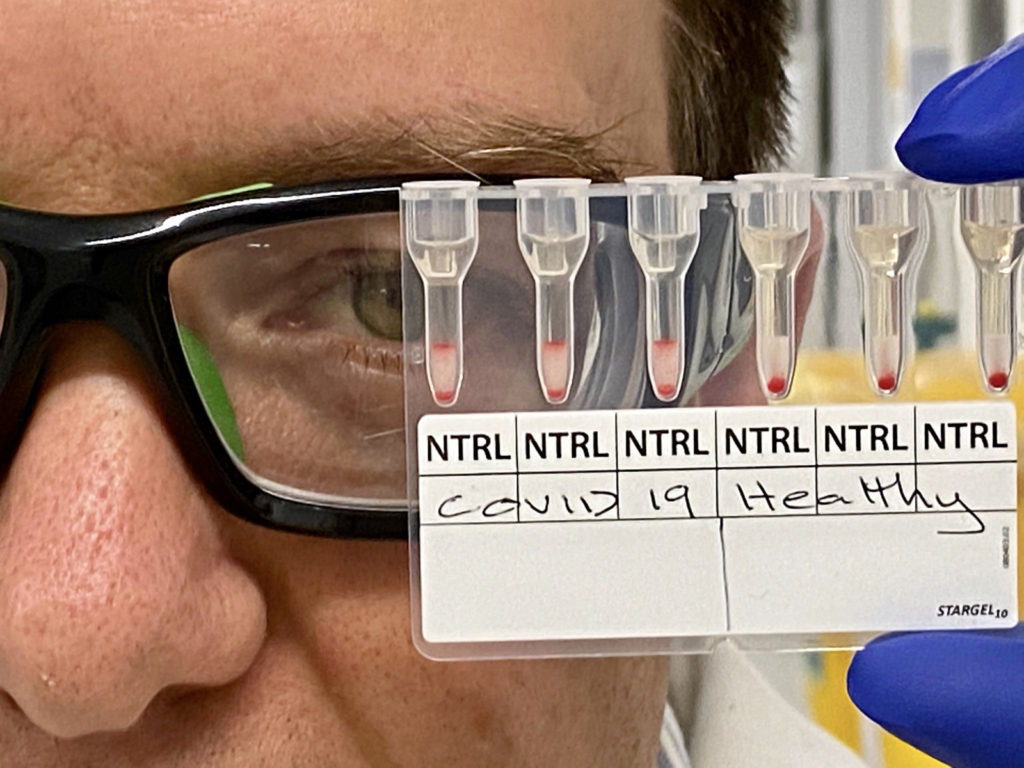
The Monash assay detects antibodies by pipetting a mixture of reagent red blood cells and antibody-containing serum onto a gel card containing separation media, incubating the card for 5 to 15 minutes, and using a centrifuge to separate the agglutinated, or clustering, cells from free cells.
“The idea behind doing that was that if we could make that work — and we still have further testing to undergo to ensure that the assay is robust and performs well in line with gold standards — then manufacturing and rolling it out would be a relatively simple task because the components are already produced in that high scale for blood typing all around the world,” Corrie said.
The assay could become a major weapon in the global fight against the pandemic, as most people who have had the virus will almost all be antibody positive by 10 to 15 days after the onset of symptoms.
“Our test then becomes useful as a surveillance and contact tracing tool. It’s also the only way of letting people know if they’ve been an asymptomatic carrier,” Corrie added.
COVID-19 and beyond
Corrie also raised the possibility of making assays that could detect other parts of the COVID-19 immune response.
“There’s some evidence that cytokine levels — these are relatively low abundance proteins in the blood — could be used to determine the severity of the disease, or to monitor the sort of cytokine storm response to that which may be associated with a severe case of COVID-19,” he said.
Beyond COVID-19 antibodies, the rapid test could be deployed to fight other diseases.
“Effectively, what we’re trying to do is take red blood cells and modify them so that they clump — agglutinate — around particular disease-related biomarkers, such as antibodies, proteins, viruses and bacteria,” he said.
“Then we can see them, especially if separated through some absorption media like a little gel column.”
That means the test could be applied to different viral infections, cancer, heart disease, sepsis or any condition in which molecular changes are indicative of disease progression.
“If we can do that in response to other biomarkers it could be really interesting,” Corrie said.
“If we can make some of these tests available outside of centralised labs, we can make them more accessible to people.”
The next step is rigorous testing and working with a commercial partner to manufacture the assay. Corrie estimates this will take an additional three to six months if the team receives further support and funding.
PPE breakthrough
Corrie’s colleague at Monash University’s Bioresource Processing Institute of Australia (BioPRIA) have also been hard at work in response to the pandemic, with chemical engineer Dr Joanne Tanner and her team tasked with solving personal protective equipment (PPE) shortages at a local level.

“Over the course of just a few weeks, a small team at BioPRIA worked with local paper and polymer manufacturers to produce and test paper-based isolation gowns for use by frontline healthcare workers,” she said.
In order to rapidly produce prototype garments, Tanner and her team measured various paper thicknesses and polymer coatings to determine the viral protection potential of the materials, as well as ensuring that they weren’t fragile or easily compromised by water.
“The material we developed passed all the standard virus protection, strength and barrier tests, and our prototype gowns ‘inspired confidence’ and were deemed ‘perfectly suitable’ for use with ‘COVID infected or at-high-risk patients’ by a healthcare professional,” she said.
“These materials could also be applied as surgical drapes, masks and other PPE where flexible, non-woven materials are required to provide short-term virus protection.”




So does this test differentiate between antibodies from other COVid infections? Or say Influenza A & B. Not only that I am a little curious that some medical practitioners state that not everyone that has COVid creates antibodies, doesn’t that infact make your test not accurate in that case?